7-month-old Korat cat was referred for a slowly-progressive
neurological disease. Circulating monocytes and lymphocytes showed the presence
of single or multiple empty vacuoles and blood leukocytes enzyme assay revealed
a very low b-galactosidase activity level (4.7 nmol/mg/hr) as compared to
unaffected parents and relatives. Histologically the cat, euthanized at the
owner request at 21 months of age, presented diffuse vacuolization and enlargement
of neurons throughout the brain, spinal cord and peripheral ganglia, severe
cerebellar neuronal cell loss, and moderate astrocytosis. Stored material
was stained with PAS on frozen sections and with RCA-I, Con-A and WGA lectins
in paraffin wax-embedded sections. Ultrastructurally, neuronal vacuoles were
filled with concentrically whorled lamellae and small membrane-bound vesicles.
In the affected cat, b-galactosidase activity was markedly reduced in brain
(18.9%) and l iver (33.25%) while total b-hexosaminidase activity showed a
remarkable increase. Quantitation of total gangliosides revealed a 3-fold
increase in brain and 1.7-fold in liver of affected cat. High-performance
thin layer chromatography (HPTLC) detected a striking increase of GM1-ganglioside.
On densitometric analysis of HPTLC bands, the absorption of GM1-ganglioside
band was 98.52% of all stained bands (GD1a ,GD1b,GT1b ). Based on clinical
onset, morphological and histochemical features, and biochemical findings,
Korat cat GM1-gangliosidosis is comparable with the human type II (juvenile)
form. However, clinical progression, survival time and level of b-galactosidase
deficiency do not completely fit with those of human type II GM1-gangliosidosis.
The disease in Korat cat is also different from other reported forms of feline
GM1-gangliosidosis. Key words Gangliosidosis G(M1) € Feline lysosomal storage
disease € Animal disease modelIntroduction
Introduction
GM1-gangliosidosis is an inherited lysosomal storage disease caused by deficient
activity of lysosomal acid b-galactosidase resulting in accumulation of GM1-ganglioside
and galactose-containing oligosaccharides in different cell types [25]. In
humans, on the basis of age of clinical onset, evolution, and clinico-pathological
findings, the disease is classified into three different forms, the infantile
(type I), juvenile (type II) and chronic or adult (Type III) forms [21, 26,
37]. Type I form is characterized by early onset, rapid progression and severe
neuroskeletal and visceral involvement. Type III form lacks of bony abnormalities
and shows slowly progressive neurological symptoms and selective glycolipid
accumulation in some brain areas [32, 40]. Type II form exhibits clinico-pathological
features falling between type I and type III GM1-gangliosidosis. Although
the clinico-pathological heterogeneity has been correlated with different
b-galactosidase gene mutations [11, 14, 38], other genetic or environmental
factors are thought to modify the phenotipic expression of GM1-gangliosidosis
[33, 39] GM1-gangliosidosis has been identified in several animal species
including cats [4, 7, 10], cattle [15], dogs [2, 27, 28], and sheep [30].
In sheep a b-galactosidase deficiency associated with a-neuraminidase deficiency
also has been reported [1]. In Korat cats another form of gangliosidosis similar
to Sandhoffąs disease (GM2-gangliosidosis type II) of humans has also been
identified [24].In this report, we describe the clinico-pathological and biochemical
features of a new form of feline GM1-gangliosidosis in a Korat cat due to
the deficient activity of b-galactosidase.
Case report
A seven-month-old, male, Korat cat was referred to the Veterinary Clinic of
the Department of Animal Pathology, University of Turin, for retarded growth
and a two weeks history of hind limb tremors and mild dyspnea. Anamnestic
data revealed regurgitation episodes. Further anamnestic investigation showed
that a kitten of a previous litter with neurological signs had died at the
age of 7 months from severe gastro-enteritis. In the last three days, the
owner reported the occurrence of seizures. Neurological examination revealed
a depressed mental status, normal cranial nerve reflexes, ataxia with hypermetria,
and spinal nerve hypereflexia. The postural reaction was normal except for
the wheelbarrowing test, which showed slow initiation and hypermetria. Ophthalmoscopic
examination showed no changes. Blood and biochemical profiles were within
normal limits. One month later, the cat was re-examinated because of progressive
paraparesis. Based on history and clinical signs a form of gangliosidosis
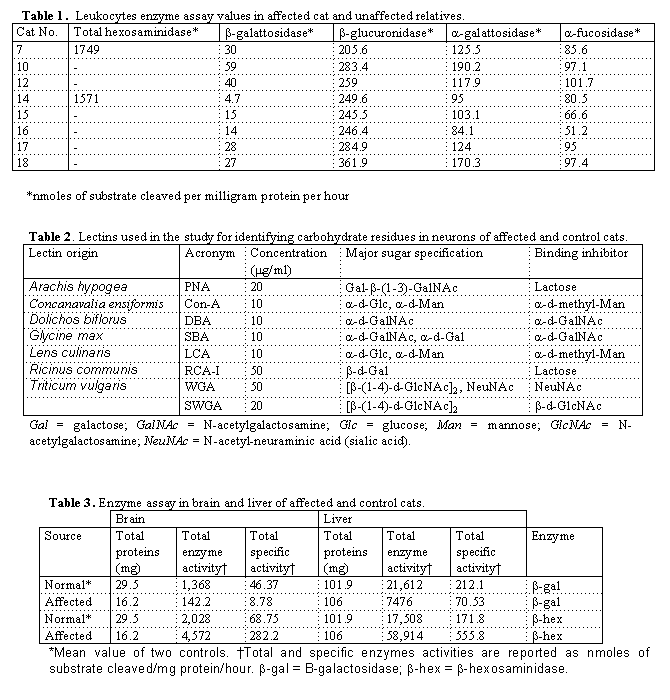
was suspected. An enzymatic assay performed on blood leukocytes revealed a
very low b-galactosidase activity level (4.7 nmol/mg/hr) in the diseased cat
while total b-hexosaminidase, b-glucuronidase, a-galactosidase and a-fucosidase
activities were within normal range, as compared to unaffected relatives (Table
1). The cat was euthanized at the owner request when he was 21 months old.
A short pedigree of affected cat is shown in Fig.1.
Material and methods
Morphological, lectin histochemical and immunohistochemical studies
White blood cells pellets from affected cat (n.14), non-affected relatives
(cat n. 12, 15, 16, 17, and 18), obligate heterozygotes (cat n. 7 and 10)
and two age-matched controls were examined on May-Grumwald-Giemsa stained
smears by light microscopy and ultrastructurally. Following post-mortem examination,
the brain was cut along the planum sagittalis into two symmetrical parts.One
hemisphere and part of spinal cord, liver, and kidney were immediately frozen
and stored at 80°C for biochemical studies. From the remaining hemisphere
and from spinal cord, liver, kidney, skeletal muscle, myocardium, and lung,
representative tissue portions were sampled and fixed in 10% neutral phosphate-buffered
formalin for histology and electron microscopy. For histological examination,
tissue samples from affected cat and from two unaffected age-matched controls
were processed routinely in paraffin wax and stained with HE, periodic-acid-Schiff
(PAS), Luxol-fast-blue (LFB) and Bodian methods. Frozen tissue sections were
also stained by PAS, oil-red-O, and Sudan black. Four mm thick, paraffin-embedded
sections from brain and spinal cord were also used for lectin histochemistry
and anti-GFAP immunohistochemistry. For lectin histochemistry, tissue sections
were incubated in a 2% H2O2 solution to block the endogenous peroxidase activity.
To reduce background staining, the sections were covered with filtered mouse
liver powder solution at a concentration of 150 mg/ml in phosphate-buffered
saline (PBS), pH 7.2, for 15 min. Sections were then incubated for 30 min
at room temperature with 8 different biotinylated lectins (Sigma-Aldrich Srl,
Milano, Italy), washed twice in PBS and covered with avidin-biotin-peroxidase
complex (ABC) (Vector Labs, Burlingame, California) for 30 min at room temperature.
An equal amount of the lectin and 0.2M of the corresponding blocking sugar
were incubated for 1 hour at room temperature before applying the solution
to the sections; this incubation served as a control for binding specificity,
and incubation of ABC alone served as a non-specific negative control. The
lectin used in this study, their acronyms, the lectin concentrations used,
their major sugar specificity, and the corresponding blocking sugars are listed
in table 2. Anti-GFAP immunohistochemistry was performed by incubating paraffin-embedded
sections with a policlonal antibody (6F2, DAKO S.p.A, Milano, Italy) 1:2000
for 45 min at 37°C after the block of endogenous peroxidase activity as above
mentioned. Sections were covered with ABC complex (Vector Labs) for 1 hour
at room temperature. For both lectin histochemistry and anti-GFAP immunohistochemistry
sections were finally incubated with a solution of 0.01% diaminobenzidine
and 3% H2O2 at room temperature for 4-8 min and lightly counterstained with
Harris haematoxylin. Positively stained structures were demonstrated by the
golden dark-brown 3.3-diaminobenzidine tetrahydrochloride-H202 reaction product.
Brain and spinal cord paraffin-embedded sections from two age-matched European
cats were used as a control for lectin histochemistry and anti-GFAP immunohistochemistry.
For ultrastructural studies tissue samples from brain, spinal cord, peripheral
ganglia, liver and kidney were post-fixed in 1% osmium tetraoxide in 0.1M
cacodylate buffer at a pH of 7.4 and stained en bloc with 5% aqueous uranyl
acetate. Blocks were dehydrated in graded ethanols solutions and embedded
in Epon/Araldite (1:1). One mm thick sections were stained with toluidine
blue and examined. Thin sections were cut 50 to 70 nm, stained by uranyl acetate
and lead citrate and observed on a Zeiss EM109 with trans-fiber-optic photography.
Leukocyte
and serum enzyme analysis
Blood samples were taken from the affected cat (n.14), unaffected relatives
(cat n. 12, 15, 16, 17, and 18), obligate heterozygotes (cat n. 7 and 10)
and two age-matched controls. Leukocytes were prepared from heparinized whole
blood. Erythrocytes were sedimented with dextran (Mr 150,000 from Sigma-Aldrich),
and the leukocytes-rich supernatant was centrifuged at 600 g for 10 min. The
pellet obtained was resuspended in distilled water for 30-40 s and NaCl was
added to the final concentration of 0.9% to lyse red blood cells. After centrifugation,
the leukocyte-rich pellet was resuspended and sonicated before enzyme assay.
The protein content of leukocyte samples was determined utilizing protein-dye
binding (Bio-Rad Laboratories, srl, Milano, Italy) [12]. The substrate for
leukocytes enzyme activity were 1.5mM of 4-methylumbrelliferyl-6-sulpho-2-acetamido-2-deoxy-b-d-glucopyranoside
(Sigma-Aldrich) for b-hexosaminidase, 1mM of 4-MU-b-galactopyranoside (Sigma-Aldrich)
for b-galactosidase, 5mM of 4-MU-b-d-glucuronide (Sigma-Aldrich) for b-glucuronidase,
and 1mM of 4-MU-a-L-fucopyranoside (Sigma-Aldrich) for a-fucosidase. Substrate
hydrolysis was measured spectrofluorometrically at an excitation wavelength
of 360 nm and an emission wavelength of 450 nm. Specific activities were reported
as nmoles of substrate cleaved per milligram protein per hour. Serum analysis
for the presence of b-hexosaminidase isoenzymes was carried out with fluorescent
staining of the bands with 2 mM 4-MU-2-acetamido-2-desossi-b-glucopyranoside
after electrophoretic separation on cellulose acetate sheets in 0.025 M citric
acid-sodium citrate, pH 5.5.
Tissue samples enzyme assay
Brain and liver samples from affected cat and two age-matched normal controls
were homogenized in a 0.25 M sucrose, 5 mM Tris, and 1mM EDTA, solution at
pH 7.5. Samples were then centrifuged at 10,000 g for 30 min at 4°C. Supernatant
was removed, aliquoted and stored at 80°C. Crude homogenate was analyzed
for total b-hexosaminidase and b-galactosidase activities with 1 mM 4-methylumbelliferyl-2-acetamido-2-deoxy-b-d-glucopyranoside
and 1mM 4-methylumbelliferyl-b-d-galactopyranoside, respectively in 0.1 M
acetate buffer, 0.125% Triton X-100, and 0.04% BSA, pH 4.4. Analysis was carried
out using a modification of Storrie and Madden method [31] at pH 4.4. Substrate
hydrolysis was analyzed spectrofluorometrically at an excitation wavelength
of 364 nm and an emission wavelength of 448 nm. Specific activities were reported
as nmoles of substrate cleaved per milligram protein per hour.
Lipids and total glycosaminoglycans (GAG) analysis
One gr of brain and liver samples from affected cat and two age-matched normal
controls were homogenized (Polytron) in 4 ml of tetrahydrofuran (Sigma-Aldrich)
and 1 ml of 10 mM phosphate buffer, pH 6.8. After centrifugation at 5000 g
for 15 min at 4°C, supernatants was mixed with ether (3:1) and centrifuged
again at 5000 g for 10 min at 4°C. Upper- and lower-phases (UP, LP) were removed
and concentrated under vacuum (rotavapor). LP was dialyzed overnight against
distilled water at 4°C, lyophilized and resuspended in chloroform/methanol
(2:1) [36]. N-acetylneuraminic acid (NANA) content of LP was determined by
the method of Svennerholm [34] and expressed as nmol/mg of wet tissue. High
performance thin-layer chromatography (HPTLC) was performed on 20 x 20-cm
silica gel HPTLC (Bracco Spa, Milano, Italy). Mobile phase was constituted
by chloroform/methanol/0.2% calcium chloride (50:42:11) for LP and chloroform/methanol/ammonia
(80:20:2) for UP . TLC of LP was developed by the resorcinol sulphuric acid
reaction [34] while TLC of UP was stained with 0.3% ninhydrin in a butilic
alcohol and 0.03% acetic acid solution. A densitometric analysis was also
carried out on HPTLC stained bands (GS-700 Imaging Densitometer, Bio-Rad Laboratories).
The relative absorption of the GM1-ganglioside band out of the total stained
ganglioside bands was expressed as a percentage. For total glycosaminoglycans
quantification, brain and liver samples were homogenized in 2 volumes of distilled
water. Proteins were digested twice by incubation on each occasion with papain
(1mg/1 ml of homogenized sample) (Boehringer Mannheim Italia S.p.A, Milano,
Italy) for 48 hours at 40°C. A few drops of octanoic acid were added to prevent
bacterial growth. After centrifugation at 12,000 g for 20 minutes, the supernatant
was brought to 7% trichloroacetic acid, held for 1 hour at 4°C and centrifuged
(12,000 g for 20 minutes). The supernatant was diluted with 4 volumes of cold
ethanol (containing 1% potassium acetate), stored overnight at -20°C and centrifuged
at 12,000 g for 10 min. Precipitated GAG were finally dissolved in distilled
water proportional to the wet weight of MV (1ml/1gr). GAG were quantified
by the carbazole method [9] using glucuronolactone as standard. The concentration
was expressed as µg of uronic acid per gr of wet tissue.
Results
Morphological and lectin histochemical studies
Light microscopic observation of leukocyte-rich pellet from the diseased cat,
revealed the presence in circulating monocytes and in some lymphocytes of
single or multiple empty vacuoles. Ultrastructurally membranebound vacuoles
were mostly empty or filled with fine fibrillo-granular material or small
vesicles (Fig. 2). Occasionally lamellated membrane structures were also observed.
Vacuoles were not present in unaffected relatives. On post-mortem examination,
no significant gross lesions, including organomegaly or skeletal abnormalities,
were found. Histological observation revealed a diffuse vacuolization and
enlargement of neurons throughout the brain, spinal cord, spinal and peripheral
ganglia. All areas in the central nervous system (CNS) were severely affected
except the granular layer of the cerebellum, ependymal and choroid plexus
cells. Severe neuronal cell loss was detected in the cerebellar cortex (Purkinje
and granular layer cells) and mid brain. Neuronal cell vacuolization was associated
in many neuroanatomic areas with moderate astrocytosis, which was readily
detected in sections immunostained for GFAP (Fig. 3). No inflammatory reaction
was observed in any part of the CNS. Affected neurons showed a marked increase
in size, a decrease in cytoplasmic staining intensity with a pale foamy or
light-pink granular appearance, frequent nuclear margination, and clear marked
cell borders. Stored material was also observed in axons. In some neurons,
Nissl substance was present in a restricted perinuclear area where in others
it was completely lost. In spinal cord some neurons showed single or multiple
large empty vacuoles. On paraffin wax-embedded sections stained with PAS the
content of vacuoles was stained only in some neurons located in medulla oblongata
and in organs and in peripheral ganglia. In liver, almost all hepatocytes,
regardless their position in the lobule showed extensive cytoplasmic vacuolization
with a large single vacuole or multiple smaller vacuoles. Foamy vacuolization
in kidney was mainly localized in the cortex with proximal and distal tubules,
and glomerular podocytes diffusely involved while cortical collecting ducts
and medullary tubular portions seemed to be unaffected. Vacuolization was
observed also in epithelial cells of large bronchi, oesophagus, and adrenal
medulla. In resin 1 mm thick sections, the content of neuronal vacuoles was
observed as blue to dark blue stained granular material (Fig. 6). No such
inclusions were seen in other cell types. Ultrastructurally, the cytoplasm
of cerebral and spinal cord neurons, cerebellar Purkinje cells, and of peripheral
ganglia were almost completely filled by membrane-bound irregularly shaped
vacuoles ranging in diameter from 0.43 to 1.9 mm in diameter (Fig. 7). In
these cells, cytoplasmic organelles were hardly observed. The stored material
inside vacuoles was most commonly composed by irregularly parallel lamellated
structures or concentrically whorled lamellae. However in largest vacuoles
smaller membrane-bound vesicles with heterogeneous content were also frequently
seen mixed with lamellated membrane structures (Fig. 7). The diameter of the
smaller vesicles ranged from 0.26 to 0.5 mm. Stored fibrillo-granular material
in hepatocytes was observed in membrane-bound vacuoles ranged 1.23 to 11.6
mm in size. Although in smaller vacuoles, the same type of stored material
was also observed in Kupfer cells. Vacuolization in renal tubular cells involved
all nephron tracts with particular intensity in proximal and distal tubules
while in the renal glomerulus, podocytes, endothelial, and Bowman's capsule
cells variable degree of cytoplasm vacuolization was found. In all kidney
affected cells, the vacuoles content was of the fibrillo-granular type.
Biochemical studies
The results of leukocytes enzyme assay on affected and unaffected relatives
are summarized in table 1. In the affected cat (n.14) a very low b-galactosidase
activity (4.7) was found. The electrophoretic analysis of b-hexosaminidase
isoenzymes revealed the presence of two bands corresponding to human hexosaminidase
A and B, in all cats tested.< The results of enzyme assay in brain and liver
of affected and control cats are summarized in table 3. Beta-galactosidase
activity in the affected cat showed a marked decrease in brain (18.9%) and
liver (33.25%) while total b-exosaminidase activity revealed a remarkable
increased activity, compared to control cats. Identification of brain total
gangliosides, as determined by the levels of lipid-bound N-acetyl-neuraminic
acid (NANA), gave a mean value of 2,600 in normal cats and 7,790 in affected
cat. In liver, total gangliosides were 100 in normal cats and 170 in affected
cat. High-performance thin layer chromatography (HPTLC) analysis showed a
striking increase of GM1-ganglioside in brain of affected Korat cat (Fig.8).
Densitometric analysis of HPTLC GM1-ganglioside bands in normal and diseased
cats was 54.55% and 98.52% respectively of the total absorption of all stained
gangliosides bands (GD1a,GD1b,and GT1b). Total GAG quantification in brain
and liver revealed a significant increase in the affected cat (147.7 and 3,816.3
respectively) compared to normal controls (29.5 and 731.42).
Discussion
The deficiency of acidic b-galactosidase and the accumulation of GM1-ganglioside
in the brain and liver of the affected cat is consistent with the diagnosis
of GM1-gangliosidosis. A preliminary analysis of the pedigree of affected
cat indicates that the disease may have been inherited as a recessive autosomal
trait since it originated with a consanguineous breeding and both parents
are phenotypically normal. One sibling may have suffered of a neurological
disease with clinical onset and features similar to those observed in the
affected cat. Enzyme activity levels determined in circulating leukocytes
from obligate heterozygotes and unaffected relatives seem not to help in identifying
carriers. Direct identification of the mutation may provide a future tool
to detect disease carrier. Although Baker et al. reported to know the occurrence
of a case of GM1-gangliosidosis in a Korat cat [ 5], biochemical and clinico-pathological
features of the disease in Korat cat were completely lacking. Based on clinical
onset, morphological features and biochemical findings, this progressive neurological
disorder seems to be comparable with human type II (juvenile) GM1-gangliosidosis.
However, clinical progression, survival time, and level of b-galactosidase
deficiency in Korat cat do not completely fit with those of human type II
GM1-gangliosidosis [18, 23, 25, 33]. As concluded for the canine models of
GM1-gangliosidosis [3], these findings indicate that it may not be completely
appropriate to apply human classification to this animal model. Although a
condition similar to human type II GM1-gangliosidosis with b-galactosidase
deficiency and GM1-ganglioside accumulation has been described in Siamese
[4] cats, significant clinico-pathological and biochemical differences are
present with the disease in the Korat cat. Clinically, Siamese cats with GM1-gangliosidosis
showed i mpaired vision, which was not evident in our case, and a more rapid
progression of the disease (death by 8-12 months) compared to the affected
Korat cat [4, 5, 6]. The longer duration of the disease may account for neuronal
loss and astrocytosis found in brain of affected Korat cat which are not reported
as characteristic in the Siamese cat model [17]. The longer duration of the
disease may also explain the higher amount of stored GM1-ganglioside in brain
associated with a higher b-galactosidase activity assayed in brain and liver
of Korat cat as compared to Siamese cats [4, 17, 19]. All these findings indicate
that GM1-gangliosidosis in Korat cat is different from that reported in other
cat breed. HPTLC and GAG quantification detected a marked storage of GM1-ganglioside
in brain and of GAG both in liver and brain indicating that enzymatic defect
involve the catabolism of lipid- and protein-bound sugars. Data obtained from
PAS reaction and lectin histochemistry on neuronal cells indicate the accumulation
of glycolipids and oligosaccharides containing a-mannose-, b-galactose-, sialic
acid-, and a-GalNAc-containing oligosaccharides [29]. All these sugar residues
are normally present in N-linked oligosaccharides and in the GM1-ganglioside
molecule [41]. Ultrastructural observations showed a clear difference in the
stored material between neuronal cells and other cell types. In neurons enlarged
lysosomes are mostly filled with lamellated structures, indicative of lipid
accumulation, while in circulating leukocytes, hepatocytes, and kidney glomerular
and tubular cells empty vacuoles or fibrillo-granular-filled vacuoles were
detected , suggestive of stored oligosaccharides or glycosaminoglycans [16].
Although gangliosides are normal constituents of the plasma membrane, the
amount and type of gangliosides in different tissue and in specific tissue
cells is variable [41]. Hence, in liver and kidney epithelial cells, stored
material is probably composed mainly of oligosaccharides rather than gangliosides
which are more abundant in neuronal cells [35]. Neuronal cell loss was particularly
severe in the cerebellum involving mainly Purkinje and granular layer cells.
It has been shown that the cellular accumulation of sphingolipids has an inhibitory
effect on protein kinase C with consequent progressive dysfunction of signal
trasduction mechanisms leading to cell death [19]. Furthermore, a recent report
has demonstrated that ceramide, which is part of the ganglioside molecule,
is involved in the triggering of cardiomyocyte apoptosis [8]. Both series
of data indicate that the accumulation of GM1-ganglioside may have initiated
multiple biochemical events leading to the neuronal cell loss observed in
affected Korat cat. Finally, lysosomal a-neuraminidase activity was not assayed
because it is highly sensitive to freezing and thawing [22], and fresh tissue
was not available in this case. Therefore a possible involvement of a-neuraminidase,
reported in other lysosomal storage diseases [1], cannot be completely excluded.
Acknowledgements
The Authors are thankful to Ms. Patrizia Morra for her precious technical
assistance. This work was supported by grants from M.U.R.S.T. (60%-1996).
References
1. Ahern-Rindell AJ, Prieur DJ, Murname RD, Raghavan SS, Daniel PF, McCluer
RH, Walkley SU, Parish SM (1988) Inherited lysosomal storage disease associated
with deficiencies of b-galactosidase and a-neuraminidase in sheep. Am J Med
Genet 31:39-56
2. Alroy J, Orgad U, Ucci AA, Schelling SH, Schunk KL, Warren CD, Raghavan
SS, Kolodny EK (1985) Neurovisceral and skeletal GM1-gangliosidosis in dogs
with beta-galactosidase deficiency. Science 229:470-472
3. Alroy J, Orgad U, DeGasperi R, Richard R, Warren CD, Knowles K, Thalhammer
JG, Raghavan SS (1992) Canine GM1-gangliosidosis. A clinical, morphologic,
histochemical, and biochemical comparison of two different models. Am J Pathol
140:675-689
4. Baker HJ, Lindsey JR, McKhann GM, Farrell DF (1971) Neuronal GM1 gangliosidosis
in a Siamese cat with b-galactosidase deficiency. Science 174:838-839
5. Baker HJ, Mole JA, Lindsey JR, Creel RM (1976) Animal models of human ganglioside
storage diseases. Fed Proc 35:1193-1201
6. Baker HJ, Reynolds GD, Walkley SU, Cox NR, Baker GH (1979) The gangliosidosis:
comparative features and research applications. Vet Pathol 16:635-649
7. Barker CG, Blakemore WF, Dell A, Palmer AC, Tiller PR, Winchester BG (1986)
GM1-gangliosidosis (type 1) in a cat. Biochem J 235:151-158
8. Bielawska AE, Shapiro JP, Jiang L, Melkonyan HS, Piot C, Wolfe CL, Tomei
LD, Hannun YA, Umansky SR (1997) Ceramide is involved in triggering of cardiomyocyte
apoptosis induced by ischemia and reperfusion. Am J Pathol 151:1257-1263
9. Bitter T, Muir HM (1962) A modified uronic acid carbazole reaction. Anal
Biochem 4:330-334
10. Blakemore WF (1972) GM1-gangliosidosis in a cat. J Comp Path 82:82:179-185
11. Boustany R-M, Qian W-H, Suzuki K (1993) Mutations in acid b-galactosidase
cause GM1-gangliosidosis in american patients. Am J Hum Genet 53:881-888
12. Bradford MM (1976) A rapid and sensitive method for the quantitation of
microgram quantities of protein utilizing the principle of protein-dye binding.
Anal Biochem 7:248-254
13. Castagnaro M, Alroy J, Ucci AA, Glew RH (1987) Lectin histochemistry and
ultrastructure of feline kidney from six different storage diseases. Virchows
Arch B 54:16-26
14. Chakraborty S, Rafi MA, Wenger DA (1994) Mutations in the lysosomal b-galactosidase
gene that cause the adult form of GMI gangliosidosis. Am J Hum Genet 54:1004-1013
15. Donnelly WJC, Sheahan BJ, Kelly M (1973) Beta-galactosidase deficiency
in GM1- gangliosidosis of Fresian calves. Res Vet Sci 15:139-141
16. Dustin P, Tondeur M, Libert J (1978) Metabolic and storage diseases. In:
Johannessen JV (ed) Electron microscopy in human medicine. McGraw-Hill Book
Co, New York, pp 149-245
17. Farrell DF, Baker HJ, Herndon RM, Lindsey JR (1973) Feline GM1 gangliosidosis:
biochemical and ultrastructural camparisons with the disease in man. J Neuropath
Exp Neurol 32: 1-18
18. Giugliani R, Coelho Dutra J, Saraiva Pereira ML, Rotta N, de Lourdes Drachler
M, Ohlweiller L, Monteiro de Pina Neto J, Pinheiro CE, Breda DJ (1985) GM1
gangliosidosis: clinical and laboratory findings in eight families. Hum Genet
70:347-354
19. Handa S, Yamakawa T (1971) Biochemical studies in cat and human gangliosidosis.
J Neurochem 18:1275-1280
20. Hannun YA, Bell RM (1987) Lysosphingolipids inhibit protein kinase C:
implications for the sphingolipidosis. Science 235:670-674
21. Kasama T, Taketomi T (1986) Abnormalities of cerebral lipids in GM1-gangliosidosis,
infantile, juvenile, and chronic type. Jpn J Exp Med 56:1-11
22. Knowles K, Alroy J, Castagnaro M, Raghavan SS, Jakowski RM, Freden GO
(1993) Adult-onset lysosomal storage disease in a Schipperke dog: clinical,
morphological and biochemical studies. Acta Neuropathol 86:306-312
23. Mutoh T, Naoi M, Takahashi A, Hoshino M, Nagai Y, Nagatsu T (1986) Atypical
adult GM1-gangliosidosis: biochemical comparison with other forms of primary
b-galactosidase deficiency. Neurol 36:1237-1241
24. Neuwelt EA, Johnson WG, Blank NK, Pagel MA, Maslen-McClure C, McClure
MJ, Wu PM (1985) Characterization of a new model of GM2-gangliosidosis (Sandhoff's
disease) in Korat cats. J Clin Invest 76:482-490
25. O'Brien JS (1983) The gangliosidoses. In: Stanbury JB, Wyngaarden JB,
Fredrickson DAS, Goldstein JL, Brown MS (eds) The metabolic bases of inherited
diseases, 5th edn. McGraw-Hill Book Co, New York, pp 592-599
26. O'Brien JS (1989) b-Galactosidase deficiency (GM1 gangliosidosis, galactosialidosis,
and Morquio syndrome type B); ganglioside sialidase deficiency (mucolipidosis
IV). In: Scriver CR, Beaudet AL, Sly WS, Valle D (eds) The metabolic bases
of inherited diseases, 6th edn. McGraw-Hill Book Co, New York, pp 1797-1806
27. Read DH, Harrington DD, Keenan TW, Hinoman EJ (1976) Neuronal-visceral
GM1-gangliosidosis in a dog with b-galactosidase deficiency. Science 194:442-445
28. Saunders GK, Wood PA, Myers RK, Shell LG, Carithers R (1988) GM1 gangliosidosis
in Portuguese water dogs: pathologic and biochemical findings. Vet Pathol
25:265-269
29. Sharon N, Lis H (1989) Carbohydrate specificity. In: Sharon N, Lis H (Eds)
Lectins. Chapman and Hall, London, pp 36-46
30. Skelly BJ, Jeffrey M, Franklin RJM, Winchester BG (1995) A new form of
ovine GM1-gangliosidosis. Acta Neuropathol (Berl) 89:374-379
31. Storrie B, Madden EA (1990) Isolation of subcellular organelles. Methods
Enzymol 182:203-220
32. Suzuki K (1991) Neuropathology of late onset gangliosidosis. A review.
Dev Neurosci 13:205-210
33. Suzuki Y, Sakuraba H, Oshima A, Yoshida K, Shimmoto M, Takano T, Fukuhara
Y (1991) Clinical and molecular heterogeneity in hereditary b-galactosidase
deficiency. Dev Neurosci 13:299-303
34. Svennerholm L (1957) Quantitative estimation of sialic acids. II. A colorimetric
resorcinol-hydrochloric acid method. Biochim Biophys Acta 24:604-611
35. Sweeley CC, Siddiqui B (1977) Chemistry of mammalian glycolipids. In:
Horowitz MI, Pigman W (eds) The glicoconjugates, vol 1. Academic Press, New
York, pp 459-540
36. Tettamenti G, Bonali F, Marchesini S, Zambotti V (1973) A new procedure
for extraction, purification and fractionation of brain gangliosides. Biochim
Biophys Acta 296:160-170
37. Warner TG, Robertson AD, O'Brien JS (1983) Diagnosis of GM1 gangliosidosis
based on detection of urinary oligosaccharides with high performance liquid
chromatography. Clin Chim Acta 127:313-326
38. Yoshida K, Oshima A, Shimmota M, Fukuhara Y, Sakuraba H, Yanagisawa N,
Suzuki Y (1991) Human b-galactosidase gene mutations in GM1-gangliosidosis:
a common mutation among Japanese adult/chronic cases. Am J Hum Genet 49:435-442
39. Yoshida K, Oshima A, Sakuraba H, Nakano T, Yanagisawa N, Inui K, Okada
S, Uyama E, Namba R, Kondo K, Iwasaki S, Takamiya K, Suzuki Y (1992) GM1 gangliosidosis
in adults: clinical and molecular analysis of 16 Japanese patients. Ann Neurol
31:328-332
40. Yoshida K, Ikeda S, Kawaguchi K, Yanagisawa N (1994) Adult GM1 gangliosidosis:
immunohistochemical and ultrastructural findings in an autopsy case. Neurol
44:2376-2382
41. Yu RK, Saito M (1989) Structure and localization of gangliosides. In:
Margolis U, Margolis RK (eds) Neurobiology of glycoconjugates. Plenum Press,
New York, pp 1-42
 Legend for figures
Legend for figures
Table 1
Fig.1 - Pedegree of Korat cat with GM1-gangliosidosis. Cat n. 11 had suffered
of an undiagnosed progressive neurological disease.
Fig.2 - Electron micrograph showing cytoplasmic membrane-bound vacuoles with
fibrillo-granular material in a circulating monocyte. Bar = 0.8 mm.
Table 2
Fig.3 - Cerebral cortex showing moderate astrocytosis. Anti-GFAP and hematoxylin
stain. Bar = 25 mm.
Fig.4 - Spinal chord. Anterior horn neurons show strong Periodic acid-Schiff
(PAS) reaction in their cytoplasms. PAS stain. Bar = 200 mm.
Fig.5 - Spinal chord. Moderate to strong cytoplasmic reaction for RCA-I lectin
is evident in the cytoplasm of many neurons. RCA-I stain. Bar = 100 mm.
Fig.6 - Cerebral cortex. Both neurons and an enlarged axon show granular intracytoplasmic
inclusions of various staining intensity. Resin-embedded one mm thick section,
toluidine blue stain. Bar = 10 mm.
Table 3
Fig. 7 - Electron micrograph showing cytoplasmic membrane-bound vacuoles with
whorled lamellated and vesicular content. Bar = 0.3 mm.
Fig. 8 - Silica gel high performance thin layer chromatography (HPTLC) of
brain gangliosides of controls (lane 1and 2) and affected Korat cat (lane
3 and 4), and standard GM1-, GM2-, and GM3-gangliosides (GM1, GM2, GM3).

